If Jonah, as the story goes, was swallowed by a fish, he might have noticed an anatomical paradox. Fish tend to swallow their prey whole, yet the biggest prey a fish can swallow is determined by the fish’s greatest width – its jaws when fully open – and not any narrower point.1 This means that prey that barely fit in the fish’s mouth (prophets included) must also pass through narrow gaps such as the opening of the intestine and the spaces between bones when the fish swallows. The solution to this apparent problem might be recognized by anyone who has tried to pack an overstuffed bag: squeezing and compression. But if fish guts are stuffed full of compressed prey, then these guts would have to be uniquely tough to handle this over-packing. After all, a ruptured gut would simply kill the fish.
A research team from the Pacific Northwest – Jaquan M. Horton and Emily Carrington of the University of Washington and the late John M. Gosline of the University of British Columbia – decided to test this expectation using intestines from Cymatogaster aggregata, the shiner perch. These little elliptical fish glint of silver in seagrasses and seaweed along sheltered shorelines. They are incredibly agile – when observed closely, they dart in all directions like Lamar Jackson on any given Sunday. But, unlike an NFL player, shiner perch don’t have a stomach; this makes them an ideal candidate to examine the ability of the intestine to compress prey and resist bursting. Since there is no wide stomach to fit the prey when first swallowed, the problem of compression must be extreme, and the intestine must be exceptionally stretchy and tough. And since the front part of the intestine (i.e., the proximal intestine) has to accommodate the initial influx of food, it should be the best at stretching and resisting high pressures before bursting. In other words, the mechanical properties of shiner perch intestine should change along its length.

To test these expectations – that shiner perch guts should be especially able to resist bursting, especially in the front part of the guts– a series of physical properties had to be measured. These began with stress and strain. Stress was the intestinal pressure the fish guts could absorb (i.e., the amount of physical force they could handle relative to their surface area), while strain was the amount the intestine deformed and stretched in response to this force. When stress is plotted against strain on a graph, the combination of force, surface area, and the distance of deformation means that the area under the graphed curve is equal to the amount of energy the intestine can absorb before bursting. In the language of physics, this is called toughness.
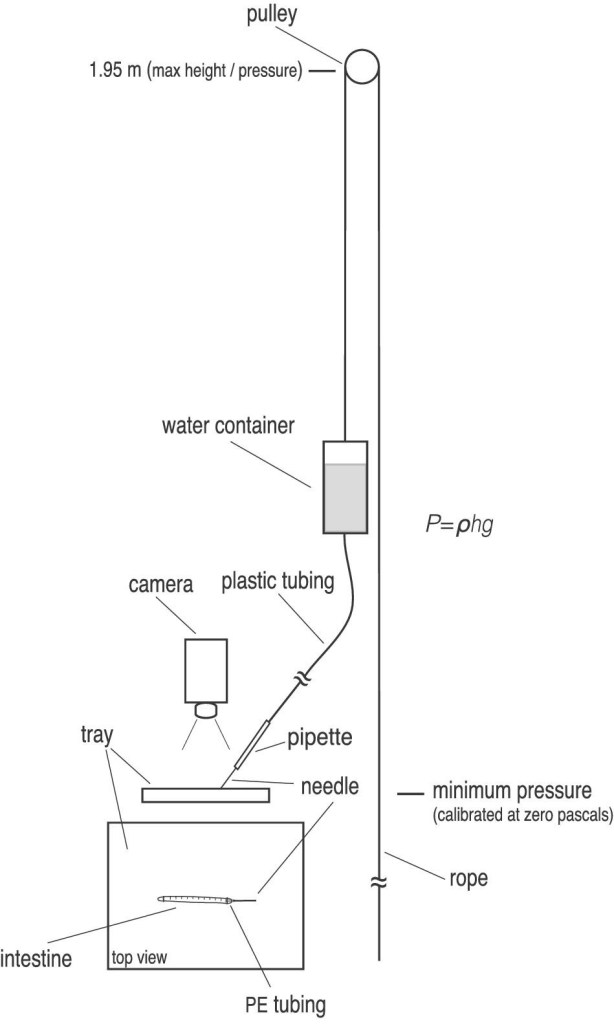
To measure these properties, Horton and colleagues used an elegant method for an inelegant end. The shiner perch intestines were stitched shut, connected to a hose, and inflated with seawater draining from a tank positioned above the intestines. Pressure on the intestines could be increased by raising the water supply further above the shiner perch guts – up until the point that these bowel balloons burst. Stress could then be calculated using the known pressure applied by the seawater, the measured radius of the intestine, and the measured thickness of its walls. Strain could be calculated by measuring how much the intestines stretched as they were inflated.
The results of this experiment aligned closely with expectations. Even from a first glance, shiner perch intestine indeed seemed especially flexible – it was able to stretch 50 percent more than human intestine. This flexibility wasn’t constant, as the fish intestine extended easily at low water pressures but became increasingly stiff as the pressure increased. The hypothesis that the front part of the guts should be best able to withstand the swallowing of large prey also stood up to scrutiny: the highest stress, strain, and toughness values were recorded in the first 25 percent of the intestine. This lines up well with the idea that, in stomachless fish, the front part of the guts replaces the stomach’s role as a temporary reserve, with the stretched-out proximal intestine forming a bulb for food storage.

So, what factors cause the high values for stress, strain, and toughness recorded in the proximal intestine? One sensible theory would be that the proximal intestine is wider than the rest of the guts or that its walls are thicker. But this is not the case: there was no meaningful difference between the width of the intestine or the thickness of its walls along the entire length of the guts. Another possibility is that the makeup (or histology) of the intestinal walls is different. The structure of the intestinal wall (and many other flexible structures found in animals) is provided by woven elastin and collagen fibres. The smooth muscles which surround and massage the intestine don’t affect the structure very much, but differences in the amount of collagen and elastin fibres and their organization are already known to alter the mechanical properties of tissues. This second solution is very likely to apply in this study of shiner perch but wasn’t directly tested. This study only examined whole, complete intestines and did not consider sections of the gut individually.

Further examination of different sections of intestine on their own could demonstrate these potential differences in intestinal wall structure and histology. Study of isolated sections of intestine could also provide further information on the mechanical properties of the guts. Since the whole intestines used in this study were inflated until they burst, the maximum values of stress, strain, and toughness were only truly recorded for the proximal intestine, since that is where the failure occurred. The values recorded for the parts of the intestine further back aren’t true maximums since they don’t reflect the highest pressure they could withstand individually; instead, they reflect the maximum pressure the intestine could withstand as a whole. However, examining individual sections of intestine would come with its own problems which this first study deliberately avoided. Slicing the intestine into sections would damage the weave of collagen and elastin fibres, alter their geometry, and ultimately reduce the strength of the tissue – meaning the maximum values for stress, strain, and toughness might not be recorded with this method either.
All-in-all, this first study of the mechanical properties of intestine in a stomachless fish tests an important hypothesis about the critical role intestine plays in compressing prey and absorbing the forces this compression produces. It also opens some exciting avenues for further research. Stomach loss in fish is surprisingly widespread: carp, minnows, pupfish, and wrasses are all stomachless. The absence of a stomach may provide advantages in regulating the acidity of the intestinal tract, increasing energy efficiency, and accommodating the chemicals released by the digestion of shells – this study begins to illuminate the necessary tradeoffs and compensation in the construction of intestines. The last point, on the digestion of shells, seems particularly intriguing. The intestines of fishes that consume hard-bodied prey (called durophages), such as animals that construct shells, must be particularly damage resistant since crushed shells have sharp edges. This problem goes beyond the mechanical properties measured in this study, since resistance to puncture from sharp objects is different than resistance to bursting from pressure.
This key step towards resolving the paradox of maximum fish prey size and the directions for future research it illuminates emphasize the importance of viewing the animal holistically. The fascinating ways fish make their living, such as predation and durophagy, don’t just depend on the swing of jaws and the snap of teeth that are so clear and compelling to human eyes. What happens inside the fish matters. The intestines have a critical role to play, especially in an animal that always bites off more than it can chew and inevitably has eyes bigger than its stomach.
1: Mihalitsis, M. & Bellwood, D. R. A morphological and functional basis for maximum prey size in piscivorous fishes. PLoS One 12, 1–19 (2017).
By Conrad D. Wilson
Conrad D. Wilson is a PhD student in the Department of Earth Sciences at Carleton University, Ottawa. His research focuses on the evolution of functional innovations in early ray-finned fishes. He can be contacted on Twitter (@conradmacwilson) or at conraddwilson@gmail.com.